Supporting Africa’s health ecosystem


In Africa, IFPMA positions our members as partners in contributing to Africa’s health ecosystem and in solving the region’s health challenges.
Overview
IFPMA and our members work in dynamic partnerships and multisectoral engagement with African public health stakeholders, policy makers, civil society, national government, and non-governmental organizations toward an innovative and sustainable health ecosystem in Africa.
We support national efforts in African countries that include advocacy to achieve universal health coverage (UHC). We also advocate for and offer solutions that build local healthcare systems through enhancing professional capacity, strengthening supply chains and procurement, financing initiatives, and increasing access to medicines and vaccines.
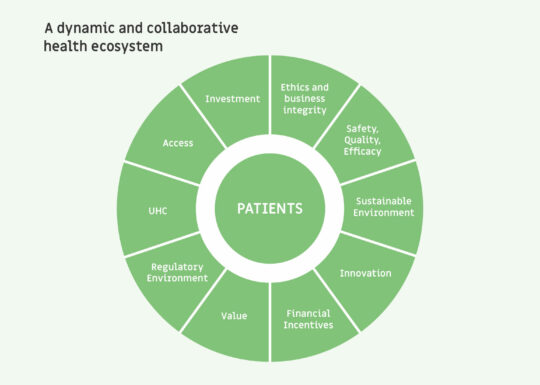
An ecosystem approach
We are working in partnership to help create a dynamic and collaborative public health ecosystem in Africa that puts people at its heart.
Our goal is to improve patient experience, health outcomes, and the allocation of resources.
We do this through an ecosystem approach that features innovation, access, health systems strengthening, regulatory harmonization and the fight against falsified medicines, UHC, investment and trade, encompassed by business integrity.
Creating a conducive policy environment
Harmonized legal and regulatory frameworks are needed to enable innovation and collaboration, as well as greater coherence between trade and health policies.
When policy environments are conducive to innovation and learning new skills, the innovative pharmaceutical industry and African health stakeholders can continue to improve population health in Africa.

Strengthening regulatory systems in Africa
IFPMA and our members believe that regulatory systems focused on Africa must be consistent and predictable to make sure people have timely access to quality medicines and vaccines with proven quality, efficacy, and safety. To support this vision, IFPMA has created a network called the Africa Regulatory Network (ARN).
Resilient health systems in Africa
Public-private partnerships play a critical role in solving global health challenges.
IFPMA engages with African policy, research, civil society, private sector, national, and public health stakeholders to strive for UHC on the continent. We understand that a multisectoral approach to collaborative innovation is necessary and partnering with private sector to expand access to vital health services is key.
As African national governments establish people-centered health services, we work to support their efforts in boosting health system resilience. We prioritize strengthening health systems by increasing equitable access to medicines for all, reinforcing capacity for vital services delivery, and propelling innovation and R&D.

Advancing universal health coverage in Africa
Universal Health Coverage (UHC) is at the heart of the health-related Sustainable Development Goals (SDGs). It is the foundation for both global health security and the health and well-being of all people throughout their lifetimes. Africa’s health systems are essential in delivering the UHC2030 action agenda. Given Africa’s population is expected to double by 2050,...
Read more
Our ethos
Ethics and integrity are cornerstones of the innovative pharmaceutical industry business culture. In Africa, they enable cross-sector collaboration to help sustain initiatives and implement innovative approaches to increasing access to quality, affordable healthcare to be guided by trust, care, fairness, respect, and honesty.
Manufacturing in Africa
IFPMA and members active on the continent, local African producers, international institutions, governments, and other stakeholders are involved in exploring solutions and initiatives to enhance and sustain local manufacturing across Africa.
of vaccine and 70% of essential medicine supply needs are met via imports in Africa
of vaccine doses needed on the continent are developed by the African vaccine manufacturing industry
of doses needed on the content should be developed, produced and supplied by African industry by 2040
Imports currently meet 99% of vaccine and 70% of essential medicine supply needs in Africa. During the COVID-19 pandemic, this major dependency, along with weakened health systems, seriously impacted the supply of vaccines and many essential medicines.
As a result, African leaders are developing the innovative pharmaceutical industry in Africa as a health security priority.

IFPMA recognizes that open dialogue between African leaders, manufacturers, suppliers, investors, international organizations, and governments is essential to develop the legal, economic, and financial frameworks to strengthen the continent’s innovative pharmaceutical industry.
Including the private sector is vital to achieve a productive national, regional, and continental market environment.
In November 2021, together with the European Federation of Pharmaceutical Industries and Associations (EFPIA), Vaccines Europe, and the European Commission, IFPMA declared our intention to consider sustainable initiatives to help stimulate production in Africa.
We keenly engage in dialogue and have led several high-level discussions, as well as supported platforms considering sustainable, market-based conditions for stronger manufacturing and partnerships in Africa.
These include with the European African Business Forum (EABF) and the Partnership for African Vaccine Manufacture (PAVM).
The Partnership for African Vaccine Manufacturing (PAVM)
Currently, the African vaccine manufacturing industry develops, produces, and supplies just 1% of vaccine doses needed on the continent.
The PAVM was established by the African Union in 2021 to work toward enabling the African industry to develop, produce, and supply over 60% of doses needed on the continent by 2040.
Since 2021, the Africa Centres for Disease Control and Prevention has worked with stakeholders across the continent to define what’s needed to develop a sustainable vaccine manufacturing industry in Africa.
European African Business Forum (EABF)
The EABF has been the main event before AU-EU Summits since 2006. It’s organized jointly by the European Commission, the African Union, and business organizations.
In 2022, an EABF Declaration representing local and international health industries operating in Africa highlighted the need for conducive business environments. These would enable public-private dialogue, better integration of EU and AU value chains, and the overall strengthening of EU-AU bilateral business relations.
Overall, there was an emphasis on the need for equal partnership between the EU and AU.
Trusted partnership and collaboration
Through partnerships with African policy makers, academia, youth innovators, health stakeholders, and the local biotechnology industry, IFPMA supports a robust research and development ecosystem in Africa.
This is growing steadily to address multidimensional challenges to accessible and quality health resources and medicines.
Our African partnerships
Our engagement with institutions
Discover Global Health Progress
See how the innovative pharmaceutical industry and our partners are achieving the Sustainable Development Goals (SDGs).